[tcb-script language=”JavaScript”]location.href=”https://drsircus.com/general/viruses-are-ph-sensitive/”[/tcb-script]
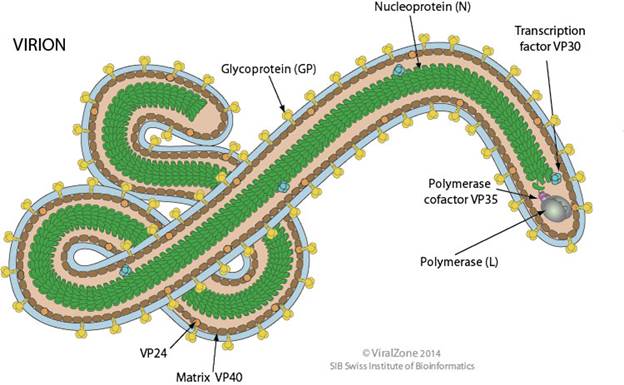 Filamentous 790 nm long for Marburg virus and 970 nm long for Ebola virus. Diameter is about 80nm.
Filamentous 790 nm long for Marburg virus and 970 nm long for Ebola virus. Diameter is about 80nm.
According to doctors at the Broad Institute of MIT and Harvard University Ebola is becoming harder to treat because rapid mutation could “render treatment and vaccines ineffective.” American scientists indicate that the initial patients diagnosed with the virus in Sierra Leone revealed more than 300 genetic modifications.
Traditional approaches to Ebola are being outclassed by the virus so we have to turn to more fundamental approaches that will stop the virus no matter what its gene sequence mutates to. Blasting Ebola with waves of alkalinity will work because many if not most viruses require a mildly acidic environment to infect the cell. They require mild acidity for maximum infectivity.
Viruses are extremely small parasitic life forms, the smallest living things on Earth. In essence, a virus is a minuscule pocket of protein that contains genetic material. Although viruses can remain dormant outside a living body, they only become active when in contact with live tissue. Once a virus infects a cell by penetrating the cell membrane, it can either lay dormant (lysogenic infection) or begin reproducing itself (lytic infection – the more common pattern). When a cell becomes full of virus, it bursts, releasing the virus to infect other host cells.
Certain viruses (including the rhinoviruses and coronaviruses that are most often responsible for the common cold and influenza viruses that produce flu) infect host cells by fusion with cellular membranes at low pH. Thus they are classified as “pH-dependent viruses.”
Fusion of viral and cellular membranes is pH dependent. “The plasma membrane of eukaryotic cells serves as a barrier against invading parasites and viruses. To infect a cell, viruses must be capable of transporting their genome and accessory proteins into the host cell, bypassing or modifying the barrier properties imposed by the plasma membrane. Entry into the host cells always involves a step of membrane fusion for enveloped animal viruses. Other enveloped viruses such as orthomyxoviruses, alphaviruses or rhabdoviruses enter the cells by the endocytic pathway, and fusion depends on the acidification of the endosomal compartment. Fusion at the endosome level is triggered by conformational changes in viral glycoproteins induced by the low pH of this cellular compartment.”[1]
In membrane biology, fusion is the process by which two initially distinct lipid bilayers merge their hydrophobic cores, resulting in one interconnected structure. The conformational transition occurs in a narrow pH range, corresponding to the optimal pH of fusion, in which the protein acquires the ability to interact with detergent micelles and lipid vesicles. This interaction leads to the insertion of the fusion peptide into the membrane, where a pore is formed. It has been suggested that the hepatitis C virus (HCV) infects host cells through a pH-dependent internalization mechanism. This HCVpp-mediated fusion was dependent on low pH, with a threshold of 6.3 and an optimum at about 5.5.[2]
When pH drops to 6 or below, rapid fusion between the membranes of viruses and the liposomes occurs. This results in the transfer of viral nucleocapsids into the liposomes. Ebola virions are taken into endothelial cells via macropinocytosis. After their formation, macropinosomes move further into the cytoplasm to acquire new markers or fuse with other vesicles of the standard endolysosomal pathway. This eventually moves the Ebola virions to more acidic compartments such as early and late endosomes that assist in the pH dependent fusion of viral and cellular membranes.[3] During this process, the cell detaches from its neighbors and loses contact with its basement membrane thanks to a mechanism of glycan mediated steric occlusion by GP.[4] The newly created particles then leave via lipid rafts, leaving a destabilized vascular system responsible for the massive blood loss characteristic of Ebola patients.[5]
Induction of Poliovirus Entry by Exposure of the Cells to Low pH
In the case of a number of enveloped viruses and diphtheria toxin, the acidic vesicles can be bypassed if cells with surface-bound virus or toxin are exposed to low pH. Under these conditions entry apparently occurs directly from the cell surface. Scientific investigation indicates that low pH is indeed required for the entry of poliovirus. The ability of cells to alter poliovirus in the presence of monensin was strongly increased at low pH. The main finding of one study is that a strain of poliovirus type 1 requires low pH for injection of its genome into the cytosol.[6]
Coronavirus infectivity is exquisitely sensitive to pH. For example, the MHV-A59 strain of coronavirus is quite stable at pH 6.0 (acidic) but becomes rapidly and irreversibly inactivated by brief treatment at pH 8.0 (alkaline). Human coronavirus strain 229E is maximally infective at pH 6.0. Infection of cells by murine coronavirus A59 at pH 6.0 (acidic) rather than pH 7.0 (neutral) yields a tenfold increase in the infectivity of the virus.
Acidic extracellular pH activates secreted lysosomal enzymes that have an optimal pH in the acidic range.[7] Hypoxia and extracellular acidity are, while being independent from each other, deeply associated with the cellular microenvironment and the spread of cancer. Intracellular pH is generally between ~6.8 and 7.4 in the cytosol and ~4.5 and 6.0 in the cell’s acidic organelles. Unlike intracellular free Ca2+ concentrations, which can rapidly change by perhaps 100-fold, pH inside a cell varies by only fractions of a pH unit, and such changes may occur quite slowly.
Inhibition of vesicular stomatitis virus (VSV) replication in LB cells by interferon (IFN) is pH sensitive. Using sensitive intracellular pH (pHi) indicators, researchers found that IFN treatment significantly raised the pHi. The increase in pHi correlated with an enhancement of the antiviral activity of IFN by primary amines. These results indicated that the IFN-induced increase in pHi may be responsible for the accumulation of G in the TGN, thereby producing G-deficient virus particles with reduced infectivity.[8]
It is the low pH inside the endosomes (pH 5–6), maintained by proton pumps within the endosomal membrane, that triggers the fusion reaction between the viral envelope and the endosomal membrane. This is a key step in the viral infection mechanism. At low pH, a major conformational change in the HA spike is induced.
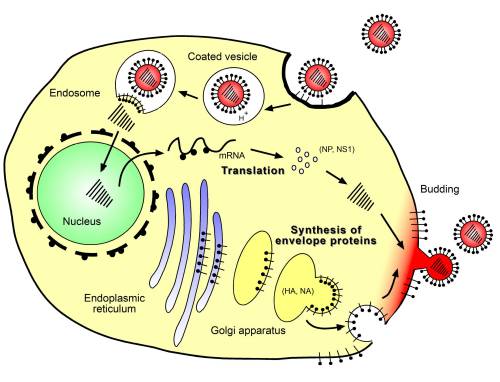
Once bound, influenza enters the host cell by endocytosis. The internalization of influenza virus is not a simple process and can be highly cell-type dependent. Viruses have been shown to enter cells by both clathrin-dependent and clathrin-independent endocytosis, as well as by macropinocytosis. Acidic environments of the endosome triggers conformational changes in HA that expose the fusion peptide, allowing for viral-endosomal fusion.[9] Exposure to low endosomal pH is also necessary for release of the individual viral ribonucleoproteins (vRNPs) from the Viruses matrix (M1) protein.
As it is with viral infections it is with cancer. The external pH of solid tumors is acidic as a consequence of increased metabolism of glucose and poor perfusion. Acid pH has been shown to stimulate tumor cell invasion and metastasis in vitro and in cells before tail vein injection in vivo.[10]
Drugs that increase intracellular pH (alkalinity within the cell) have been shown to decrease infectivity of pH-dependent viruses. However pharmaceutical drugs that do this can provoke negative side effects. Sodium bicarbonate is the best way to increase pH in clinical emergency conditions and has been known as far back as the Spanish Flu pandemic of 1918 to save lives.
The proven value of Arm & Hammer Pure Bicarbonate of Soda as a therapeutic (healing) agent is further evinced by the following voluntary testimony of Edward R. Hays, M.D. , in his latter no the Church & Dwight Company:
In 1918 and 1919 while fighting the ‘Flu’ with the U.S. Public Health Service it was brought to my attention that rarely anyone who had been thoroughly alkalinized with Bicarbonate of Soda contracted the disease, and those who did contract it, if alkalinized early, would invariably have mild attacks. I have since that time treated all cases of “Cold,” Influenza and LaGripe by first giving generous doses of Bicarbonate of Soda, and in many, many instances within 36 hours the symptoms would have entirely abated.
Further, within my own household, before Woman’s Clubs and Parent-Teachers’ Associations, I have advocated the use of Bicarbonate of Soda as a preventive for “Colds,” with the result that now many reports are coming in stating that those who took “Soda” were not affected, while nearly everyone around them had the “Flu.”
“Besides doing good in respiratory affections, Bicarbonate of Soda is of inestimable value in the treatment of Alimentary Intoxication, Pyelitis (inflammation of the pelvis), Hyper-Acidity of urine, uric acid disturbance, rheumatism and burns. An occasional three-day course of Bicarbonate of Soda elimination increases the resisting power of the body to all Infectious Diseases.
Dr. Volney S. Cheney reported[11] that, “A number of cases of colds of varying severity were carefully studied in the laboratory. Observation was made as to the degree of acidity of the urine; the CO2, combining power of the blood as an indicator of the alkaline reserve; tests were also made to determine the calcium content of the blood, the sugar content; non-protein nitrogen and the basal metabolism. The urine invariably carried a higher degree of acidity than the normal-in some cases as high as 800 (normal 350); the CO2 combining power of the blood in all cases was low, the highest being 52 per cent; the sugar content of the blood was generally decreased (below 100 mg. per 100 c.c.); the metabolic rate was always on the minus side. (These cases were carefully selected because of their lack of any symptoms of disturbed thyroid activity.) There is a change in the blood chemistry and, consequently, there must be a change in the tissues supplied by the blood. There is a decrease in the bicarbonates or reserve bases contained in the blood plasma and the tissues. These findings seem to point the way to the conclusion that a cold is a disturbance of the alkaline balance or reserve, in other words, a mild acidosis, or perhaps better stated, a lessening of the “buffer” action of the blood plasma through a decrease in its bicarbonate content.”
Sodium bicarbonate is the important medicine because it gives more carbon dioxide to the body and especially the blood in the form of bicarbonates. Bicarbonate in the blood is easily turned into carbon dioxide (CO2) and the reverse is true in biochemical reactions that happen almost at the speed of light. The bottom line to what happens when one takes sodium bicarbonate orally is that it turns to CO2 in the stomach driving bicarbonates into the blood, which helps more blood and oxygen delivery to the cells.
After doing his experiments, Dr. Cheney reported: “I have been able to induce all the symptoms of a cold, in varying degree from a simple coryza to that of la grippe and the “flu,” by the induction of an artificial acidosis through the administration of ammonium and calcium chlorides. The degree of severity of the symptoms was in direct ratio to the degree of acidosis induced. In the more severe degree of acidosis, all the classical symptoms of the “flu” were present, even including a low degree of fever. The symptoms rapidly subsided upon the administration of sodium bicarbonate in large doses by mouth and by rectum.
Sodium Bicarbonate is one of the most flexible medicinals in terms of methods and modes of administration. It can be injected in emergency room situations, taken orally, nebulized, used transdermally as a lotion or paste, put in enemas and in larger quantities in therapeutic baths. Just dissolve it in water or when treating cancer it can also be mixed with Blackstrap Molasses, maple syrup, or a good honey as well as with lemon or mix with citric acid for use in baths or when making your own bath bombs.
There is no question that plasma bicarbonate concentrations are shown to increase after oral ingestion. The most important effect of bicarbonate ingestion is the change in acid-base balance in biological fluids. In Europe, spa-goers drink bicarbonate-rich water to heal ulcers, colitis and other gastric disorders. Ingesting bicarbonate by way of bathing stimulates circulation, possibly benefiting those with high blood pressure and moderate atherosclerosis. It would be highly negligent to exclude it from Ebola treatments.
Increases of Carbon Dioxide and Bicarbonates Lead to Increased Oxygen
The most important factor in creating proper pH is increasing oxygen because no wastes or toxins can leave the body without first combining. with oxygen. The more alkaline you are, the more oxygen your fluids can hold and keep. Oxygen also buffers/oxidizes metabolic waste acids helping to keep you more alkaline. “The Secret of Life is both to feed and nourish the cells and let them flush their waste and toxins”, according to Dr. Alexis Carrell, Nobel Prize recipient in 1912. Dr. Otto Warburg, also a Nobel Prize recipient, in 1931 & 1944, said, “If our internal environment was changed from an acidic oxygen deprived environment to an alkaline environment full of oxygen, viruses, bacteria and fungus cannot live.”
The position of the oxygen disassociation curve (ODC) is influenced directly by pH, core body temperature and carbon dioxide pressure. According to Warburg, it is the increased amounts of carcinogens, toxicity and pollution that cause cells to be unable to uptake oxygen efficiently. This is connected with over-acidity, which itself is created principally under low oxygen conditions.
According to Annelie Pompe, a prominent mountaineer and world-champion free diver, alkaline tissues can hold up to 20 times more oxygen than acidic ones. When our body cells and tissues are acidic (below pH of 6.5-7.0), they lose their ability to exchange oxygen, and cancer cells love that.
Special Note: This is not the only way to skin the cat (virus). Directly supporting the immune system through a number of natural means and replenishing Vitamin C faster than Ebola strips it from the body creating lightening Scurvy and massive hemorrhage is another. Hitting the body hard with Glutathione and selenium is yet another potent and intelligent avenue of treatment that is not being pursued by the western medical establishment that prefers to complain that there is no treatment.
My book Sodium Bicarbonate is available in hardcopy as well as in eBook forms.


[1] Viral membrane fusion: is glycoprotein G of rhabdoviruses a representative of a new class of viral fusion proteins? Braz J Med Biol Res ;vol.38 no.6; Ribeirão Preto June 2005; http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-879X2005000600002
[2] Hepatitis C virus glycoproteins mediate low pH-dependent membrane fusion with liposomes; Lavillette D et al; J Biol Chem. 2006 Feb 17;281(7):3909-17. Epub 2005 Dec 15; http://www.ncbi.nlm.nih.gov/pubmed/16356932
[3] Saeed, M.F., Kolokoltsov, A.A., Albrecht, T. & Davey, R.A. (2010). Cellular Entry of Ebola Virus Involves Uptake by a Macropinocytosis-Like Mechanism and Subsequent Trafficking through Early and Late Endosomes. PLOS Pathogens, 6(9).
[4] Francica, J.R., Varela-Rohena, A., Medvec, A., Plesa, G., Riley, J.L. & Bates, P. (2010). Steric Shielding of Surface Epitopes and Impaired Immune Recognition Induced by the Ebola Virus Glycoprotein. PLOS Pathogens, 6(9), e1001098.
[5] Bavari, S., Bosio, C.M., Wiegand, E., Ruthel, G., Will, A.B., Geisbert, T.W., Hevey, M., Schmaljohn, C., Schmaljohn, A. & Aman, M.J. (2002). Lipid Raft Microdomains: A Gateway for Compartmentalized Trafficking of Ebola and Marburg Viruses. The Journal of Experimental Medicine, 195(5), 593-602
[6] Mechanism of Entry into the Cytosol of Poliovirus Type 1 : Requirement for Low pH’ INGER HELENE MADSHUS et al; Published April 1, 1984; http://jcb.rupress.org/content/98/4/1194.full.pdf
[7] Acidic extracellular microenvironment and cancer; Yasumasa Kato; Cancer Cell International;2013, 13:89;doi:10.1186/1475-2867-13-89; http://www.cancerci.com/content/13/1/89
[8] Primary amines enhance the antiviral activity of interferon against a membrane virus: role of intracellular pH; Maheshwari RK et al; J Gen Virol.; 1991 Sep;72 ( Pt 9):2143-52. http://www.ncbi.nlm.nih.gov/pubmed/1654374
[9] Viruses 2012, 4, 1144-1168; doi:10.3390/v4071144
[10] Bicarbonate Increases Tumor pH and Inhibits Spontaneous Metastases; Ian F. Robey,1; Cancer Res 2009; 69: (6). March 15, 2009; http://www.czlonkamediagroup.com/files/bicarbonate-increases-tumor-ph-and-inhibits-metastases.pdf
[11] “The Common Cold” Etiology, Prevention and Treatment*VOLNEY S. CHENEY, M. D., FELLOW A. P. H. A.Medical Director, Armour and Co., Chicago,Ill. http://ajph.aphapublications.org/cgi/reprint/18/1/15.pdf
What’s Happening
- Jan 24: Treatment Recommendations For New Virus That Is Shutting Down Entire Cities
- Jan 27: Coronavirus Epidemic – Should We Panic Yet?
- Jan 28: Coronavirus Killing Even Healthy People?
- Jan 29: The Joke of Medical Quarantines
- Jan 30: Navigating Viral Storms and Avoiding Death
- Jan 31: Dosages and Treatments for Coronavirus Infections
- Feb 3: Glutathione Against the Coronavirus
- Feb 6: They Don’t Want You to Know Basic Viral Treatments – Water is the Most Basic Treatment
- Feb 10: World Ready to Freak?
- Feb 13: World Health Experts Have It Wrong
- Feb 17: Deadly Viral and Economic Reapers
- Feb 19: Mild Mannered Coronavirus
- Feb 24: The World Is Running Out Of Time
- Feb 28: Virus, 5G and Pollution Combine to Destroy Wuhan
- Mar 2: Untrustworthy Coronavirus Tests and Statistics
Dr.Sircus is a reader-supported publication. To receive new posts and support my work, consider becoming a free or paid subscriber.
Subscribe now



comments