Sodium, potassium, and magnesium bicarbonate are miracle medicines not because of any mumbo jumbo but because of hard medical science, medical history, and basic physiology. It continues to be a medical tragedy that the least expensive, most widely available safe, and effective medicine is overlooked.
However, no matter what anyone says or thinks about sodium bicarbonate, simple baking soda is still used in ICU departments, emergency rooms, and ambulances to save lives every day. The urge to give bicarbonate to patients should be irresistible, even to cancer and diabetic patients who need it the most.
Sodium bicarbonate (NaHCO3) is a natural compound found throughout nature—in the ocean, in the soil, in our foods, and our bodies. Baking soda is a neutralizer of many other compounds, making it extremely helpful as a medicine in this age of toxicity that we are all presently living in.
Its backbone characteristic is to maintain the balance of carbon dioxide, bicarbonates, and pH levels in the blood. CO2 levels in the blood, which is increased by sodium bicarbonate intake, are vital to the cells’ oxygen delivery. Something as simple as baking soda can give almost instant relief for a wide range of medical problems because of its ability to raise both oxygen levels and cell voltage. Both are measured and changed by pH levels, which are easily adjusted with bicarbonates’ oral intake.
Sodium bicarbonate is serious lifesaving medicine extremely helpful when treating cancer, diabetes, sepsis, neurological diseases, and even bacterial, fungus, and, most importantly, viral infections. Most viruses are pH-sensitive. For the majority, a drop in pH serves as a trigger for host penetration. Some bacterial toxins also rely on low pH as a cue. The existence of acidosis in the blood simplifies viruses’ entry to host cells that then thrive and replicate.
Some think that baking soda is the most potent anti-fungal substance because of its ability to rapidly change Candida colonies’ pH from acid to alkaline, which effectively kills off the yeast before it has time to adapt to new environments. The problem with anti-fungal drugs is that fungi are extremely adaptive and can accommodate themselves to a new environment in three to four days, which renders anti-fungal drugs largely ineffective. The fungi do not adapt to the baking soda. Nor do bacteria, fungus, and viruses adapt to iodine, another strong natural anti-infectious agent.
Science Daily said, "Infectious diseases are the world’s number-one cause of death, with pathogenic fungi being responsible for extremely dangerous infections. Worldwide, more than €6 billion are spent each year on anti-fungal medications, and the total costs of the medical treatment of infectious diseases caused by pathogenic fungi are estimated in the order of hundreds of millions of Euros."[1]
In terms of importance, appropriate bicarbonate levels are just as crucial as hydration, proper breathing (getting enough oxygen), staying warm, maintaining healthy pH (high oxygen status), and optimal nutrition.
Dr. Lynda Frassetto of the University of California, San Francisco, says, "Insufficient amount of bicarbonates in our blood reduces our capabilities to manage (neutralize and dump) the acid our body produces. This is the cause of aging. The age of 45 is the average age when human beings start to show symptoms of diabetes, hypertension, osteoporosis, and many other adult degenerative diseases. And since we cannot manage the acid, we accumulate acidic wastes in our body. These wastes show up as cholesterol, fatty acid, uric acid, urate, sulfate, phosphate, kidney stones, etc."[2]
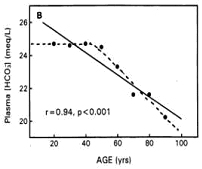
Dr. Frassetto attributes the reduction of bicarbonates in the blood as we age to generations of modern American diets. She also points out in another paper that the average diet today is high in sodium and low in potassium, contrary to the human body’s original genetic makeup.[3]
It is this reduction of bicarbonates in the blood that affects blood flow. It makes it difficult to manage the continuous outpour of acid, making it difficult to eliminate acid waste from the body. Therefore many acid-induced degenerative diseases such as blood clots, acid reflux, heart disease, osteoporosis, gout, diabetes, high blood pressure, kidney disease, cancer, strokes, etc. Alzheimer’s disease is nothing but slow acidification of the brain. All these diseases are caused by systemic acidosis, which means insufficient bicarbonates in the blood.
In Love with CO2 and Bicarbonates
Our life and health depend on having enough CO2 in the blood and tissues. Carbon dioxide is a nutrient and a product of respiration. Its lack or deficiency is a starting point for many disturbances in the body.
It is extremely important to note that CO2 is bicarbonate’s twin sister. When we take bicarbonate, the acid in the stomach or lemon in the glass turns bicarbonates into CO2. In the blood, we see a constant and ultra-fast interaction between bicarbonate and CO2. To understand bicarbonate medicine and why it is as powerful and important as it is, one must appreciate CO2 physiology.


Public opinion tends to think of carbon dioxide as a waste product or even a poison. (It is sometimes confused with carbon monoxide, which is a poison). In the 19th century, Zuntz, in Berlin, recognized that carbon dioxide, unlike oxygen, is not carried by hemoglobin. He showed that carbon dioxide is combined with bases, chiefly as sodium bicarbonate, which plays a part in acid-alkaline balance in the blood. Most of the carbon dioxide is dissolved in the plasma, both in simple solution and that combined with alkali into the bicarbonates.
In medicine, up to 5% carbon dioxide is added to
pure oxygen for stimulation of breathing after
apnea and to stabilize the O2/CO2 balance in blood.
Carbon dioxide is transported in the blood in three forms: dissolved in plasma, as bicarbonate, and coupled to proteins in the form of carbamino compounds. Bicarbonate represents the largest fraction of the CO2 in the blood (~88%). The bicarbonate buffer system is an acid-base homeostatic mechanism involving the balance of carbonic acid (H2CO3), bicarbonate ion (HCO−), and carbon dioxide (CO2) to maintain pH in the blood and duodenum, among other tissues, to support proper metabolic function.
Catalyzed by carbonic anhydrase, carbon dioxide (CO2) reacts with water (H2O) to form carbonic acid (H2CO3), which in turn rapidly dissociates to form a bicarbonate ion (HCO−) and a hydrogen ion (H+).
Under clinical conditions, low oxygen and low carbon dioxide generally occur together. Therapeutic increase of carbon dioxide, by inhalation of this gas diluted in air, is often an effective means of improving the oxygenation of the blood and tissues.[4] In the presence of a large amount of carbon dioxide, the hemoglobin molecule changes its shape slightly in a way that favors the release of oxygen.
Carbon dioxide is one of the most important gases for life. It is healthy and extremely necessary. CO2 is good, and without enough of it, we get sick. CO2, the waste product of cell metabolism, is not waste at all. Plants thrive on it, and our lives depend on it.
Dr. Konstantin Buteyko said, "CO2 is the main source of nutrition for any living matter on Earth. Plants obtain CO2 from the air and provide the main source of nourishment for animals, while both plants and animals are nourishment for us. The great resource of CO2 in the air was formed in pre-historical times when the amount was about 10%."[5]
CO2 – Alkalinity – Bicarbonate
Life on Earth depends on appropriate pH levels in and around living organisms and cells. Human life requires a tightly controlled pH level in the serum of about 7.4 (a slightly alkaline range of 7.35 to 7.45) to survive. "When your body fluids contain too much acid, it is known as acidosis. Acidosis occurs when your kidneys and lungs cannot keep your body’s pH in balance. The body has to produce bicarbonates to neutralize acids, but as we get older, live in toxic environments, take too many medications and eat the wrong foods, we fall behind. Many of the body’s processes produce acid.
Sodium bicarbonate is indicated in the treatment of metabolic acidosis, which may occur in severe renal disease, uncontrolled diabetes, circulatory insufficiency due to shock or severe dehydration, extracorporeal circulation of blood, cardiac arrest, and severe primary lactic acidosis. Sodium bicarbonate is also indicated in treating certain drug intoxications, including barbiturates. Sodium bicarbonate treats severe diarrhea, which is often accompanied by a significant loss of bicarbonate.
Drs. Jeffrey Kraut & Nicolaos Madias write, "Acute metabolic acidosis is associated with increased morbidity and mortality because of its depressive effects on cardiovascular function, facilitation of cardiac arrhythmias, stimulation of inflammation, suppression of the immune response, and other adverse effects."[6]
Drs. Sandra Sabatini and Neil A. Kurtzman say, "A fall in extracellular and intracellular pH can affect cellular function via different mechanisms, and treatment should be directed at improving both parameters. In addition to supportive measures, treatment has included administration of base, primarily in the form of sodium bicarbonate."[7] So it is more than sad that bicarbonate administration’s utility to patients with severe metabolic acidosis remains controversial even though that is exactly what bicarbonates do, neutralize acids.
Since our body is about 80 to 99% water (depending on how you count), consuming bicarbonates and magnesium will directly affect the vast majority of our bodies. Dr. Gerald Pollack is a Professor of Bioengineering at the University of Washington. He says that the previous 60 to 70 percent estimate of the human body’s water content was based on the volume of water in the body as a percentage of the body’s total volume. Pollack made his revised estimate based on the percentage of individual water molecules in the body, compared to the total number of molecules. By the molecule method of calculation, the human body’s water percentage becomes a whopping 99%. Pollack’s assertion that water is more important to the human body than previously realized supports Sharon Kleyne’s theory that the lack of adequate water in the body, also known as "dehydration," is ultimately responsible for most disease processes. Kleyne and Pollack agreed that it makes little sense to study disease and body functioning without water in the equation.[8]
So treating water makes sense and the absolute best way is to drink magnesium bicarbonate water. The second best way is with an inexpensive product called pH Adjust, which is a combination of sodium bicarbonate, potassium bicarbonate, and magnesium carbonate. Ingesting bicarbonates and magnesium with water is the simplest way to achieve an ideal pH balance in the body. It is important to note that adding these minerals will make your water alkaline in a far better way than any alkaline water machine will that create high pH water with little alkalinity.
When we take bicarbonates:
- Increases elimination of toxins and wastes
- Increases absorption of nutrients
- Acts as a scavenger of free radicals
- Reduces inflammation
- Helpful with skin conditions
- Increases CO2 and bicarbonate concentrations in the blood.
- Increases Oxygen Carrying Capacity and Delivery
- Anti-Aging
- Excellent Kidney Health
- Preventive Cancer Treatment
Drinking high alkaline magnesium/bicarbonate water increases digestion and nutrient absorption, increasing energy levels as well as detoxification of the cells. One of the main functions of water is to carry harmful acidic waste products; but, with the accumulation of acidic waste from polluted air, water, and food, combined with the normal toxins created by the metabolic processes of the human body, we are finding it harder and harder to maintain the proper pH balance for health.
This is the introduction to the 3rd edition of my Sodium Bicarbonate book.
Special Note: I have developed a full system of natural medicine that I call Natural Allopathic Medicine. At heart, it transposes ICU use of the above natural drugs (and more than several others) used in dire medical situations to home use for acute and chronic illnesses, including cancer. Instead of limiting their usefulness for near-death cases, we employ them at high dosages daily to reverse disease. I do consultations from a distance to teach people how to set up intensive treatment centers in their own homes. Much of the information can also be gathered freely on my site or in my many ebooks, books in print, and books in other languages.
[1] Molecular causes for life-threatening fungal infections in case of sepsis unraveled
July 27, 2012. Source: Medical University of Vienna https://www.sciencedaily.com/releases/2012/07/120727082518.htm
[2] Journal of Gerontology: BIOLOGICAL SCIENCES, 1996, Vol. 51A. No. 1, B91-B99
[3] European Journal of Nutrition, Vol. 40, Number 5 (2001). �Steinkopff Verlag 2001
[4] Henderson, Y. Carbon Dioxide. Article in Encyclopedia of Medicine. 1940
[5] https://buteyko.co/pages/health-benefits-of-co2
[6] John P. Cunha, Medical and Pharmacy Editor. Sodium Bicarbonate, https://www.rxlist.com/consumer_sodium_bicarbonate_baking_soda/drugscondition.htm, Copyright © 2020 by RxList Inc
[7] Kraut, J., Madias, N. Treatment of acute metabolic acidosis: a pathophysiologic approach. Nat Rev Nephrol 8, 589–601 (2012). https://doi.org/10.1038/nrneph.2012.186.
[8] https://www.prweb.com/releases/2014/03/prweb11623245.htm Visited 1/15/2021
Dr.Sircus is a reader-supported publication. To receive new posts and support my work, consider becoming a free or paid subscriber.
Subscribe now




comments