This informal video of physicist and distinguished nuclear expert Dr. Chris Busby has him recommending magnesium and calcium supplementation to everyone concerned about the radiation exposure from Fukushima. Nuclear particles do have a strong propensity to bind with DNA and that is why we see cancers develop, even after low exposure, and that is also why we see drops in fertility and the onset of sterility.
Dr. David Brownstein and I have made a similar universal call for iodine in that the nutritive type of iodine protects the thyroid from the radioactive type of iodine. One has to be a fool not to be taking iodine because 95 percent of us are deficient in iodine, thus making our thyroids sitting ducks (sponges) for the radioactive type. A thyroid deficient in iodine becomes like an intense vacuum cleaner hungry for anything even looking like a halogen (fluoride, bromide, chlorine, rocket fuel). Because so much blood goes through the thyroid it has a fantastic capacity to concentrate radioactive iodine and these halogens no matter how low the concentration is of them in the blood.
Exposure to radiation causes a cascade of free radicals that wreak havoc on the body. Radiation decimates the body’s supply of glutathione.
We are even greater fools though if we are not taking magnesium with an equal amount of calcium (though most often that equal amount of calcium is already supplied by the diet) as the radioactive clouds and rain circle the northern half of the globe. Though I would say the same for iodine and selenium (selenium is not only essential for glutathione production but is also important to take with iodine), magnesium and calcium will protect us and our children from some of the nastiest long-term effects of radiation exposure.
Without sufficient magnesium, the body accumulates toxins and acid residues, degenerates rapidly, and ages prematurely.
“Depleted uranium (DU) is highly toxic to humans, both chemically as a heavy metal and radiological as an alpha particle emitter,” writes Dr. Rosalie Bertell, Canadian Epidemiologist.[1] A new study, conducted by biochemist Dr. Diane Stearns at Northern Arizona University confirms that, separate from any radiation risks, cells exposed to uranium will bond with the metal chemically.[2] Uranium and phosphate have a strong chemical affinity for each other and the DNA and mitochondria are loaded with phosphate so uranium is a DNA and mitochondria deep-penetration bomb.
Magnesium and DNA
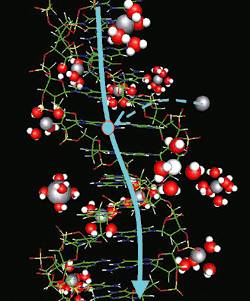
Mechanism of electric conductivity in DNA. Magnesium (silver circles) with no surrounding water supplies holes (light blue circles) to the DNA, which is an insulator. The supplied holes move along the DNA (light blue line).
Magnesium ions play critical roles in many aspects of cellular metabolism. They stabilize structures of proteins, nucleic acids, and cell membranes by binding to the macromolecule’s surface and promote specific structural or catalytic activities of proteins, enzymes, or ribozymes. Magnesium is required as a cofactor for many enzyme systems. It is required for protein synthesis and for both anaerobic and aerobic energy generation and for glycolysis, either indirectly as a part of magnesium-ATP complex, or directly as an enzyme activator. Magnesium has a critical role in cell division. It has been suggested that magnesium is necessary for the maintenance of an adequate supply of nucleotides for the synthesis of RNA and DNA.
Magnesium plays a critical role in vital DNA repair proteins. Magnesium ions’ synergetic effects on the active site geometry may affect the polymerase closing/opening trends.
Distinct structural features of DNA, such as the curvature of dA tracts, are important in the recognition, packaging, and regulation of DNA. Physiologically relevant concentrations of magnesium have been found to enhance the curvature of dA tract DNAs.
Dr. Paul Ellis informs us that, “Magnesium ions are central to the function of the DNA repair proteins, apurinic/apyrimidic endonuclease 1 (Ape1) and polymerase A (Pol A). These proteins are key constituents of the base excision repair (BER) pathway, a process that plays a critical role in preventing the cytotoxic and mutagenic effects of most spontaneous, alkylation, and oxidative DNA damage.”[3]

A DNA polymerase is an enzyme that assists in DNA replication. Such enzymes catalyze the polymerization of deoxyribonucleotides alongside a DNA strand, which they “read” and use as a template. The newly-polymerized molecule is complementary to the template strand and identical to the template’s partner strand. DNA polymerase is considered to be a holoenzyme since it requires a magnesium ion as a co-factor to function properly. DNA-polymerase initiates DNA replication by binding to a piece of single-stranded DNA. This process corrects mistakes in newly-synthesized DNA.
Single-stranded RNA can be highly stabilized by magnesium ions.
The interaction of nucleic acids with magnesium plays an important role in stabilizing their tertiary structure. Site-specific binding of magnesium to nucleotide bases, particularly guanine, has been observed in certain crystal structures. Magnesium, an essential metal that is important in the normal functioning of DNA alters the tumorigenic [tumor-causing] process when it interacts with heavy metals, which are damaging DNA molecules.[4]
Calcium
The recommended daily allowance (RDA) of calcium is 1,000 mg per day for men and 1,500 mg for women.
A healthy cell has high magnesium and low calcium levels. The higher the calcium level and the lower the magnesium level in the extra-cellular fluid, the harder is it for cells to pump the calcium out. The result is that with low magnesium levels the mitochondria gradually calcify and energy production decreases. Up to 30% of the energy of cells is used to pump calcium out of the cells so we really need to be careful and make sure we are taking enough magnesium.
Magnesium and calcium share a common route of absorption in the intestinal tract and appear to have a mutually suppressive effect on each other. If calcium intake (or dairy intake) is unusually high, calcium will be absorbed in preference to magnesium. Coronary artery calcification is common, severe and significantly associated with ischemic cardiovascular disease in adult end-stage renal disease patients.[5] The amount of calcium in the coronary arteries reliably predicts heart attack risk and is measured by what is called one’s calcium score. UCLA cardiologist, Dr. Matt Budoff, a long-time champion of the Coronary Calcium Scan and author of the AHA paper, says, “The total amount of coronary calcium (Agatston score) predicts coronary disease events beyond standard risk factors.”
Magnesium and calcium work together to control muscle action though calcium becomes increasingly toxic in the face of any kind of magnesium deficiency. Magnesium and calcium are paired minerals and it is vital we take into account the balance between them. Several studies have reported that increasing calcium in the diet significantly reduces the absorption of magnesium. Calcium intakes above 2.6 grams per day may reduce the uptake and utilization of magnesium by the body and excessive calcium intakes increase magnesium requirements. So much stress is placed on the importance of calcium that we may, in fact, be taking in too much and harming magnesium absorption.
According to Dr. Sarah Mayhill, “Calcium and magnesium compete for absorption and so too much calcium in the diet will block magnesium absorption. Our physiological requirement for calcium to magnesium is about 2:1. In dairy products the ratio is 10:1. So, consuming a lot of dairy products will induce a magnesium deficiency.”
Magnesium taken in proper dosages can solve the problem of calcium deficiency. – Dr. Nan Kathryn Fuchs
Another reason Dr. Busby was on solid medical ground in recommending magnesium is that it is also vital in glutathione synthesis. Glutathione synthetase requires ?-glutamyl cysteine, glycine, ATP, and magnesium ions to form glutathione.[6] In magnesium deficiency, the enzyme y-glutamyl transpeptidase is lowered.[7] Data demonstrates a direct action of glutathione both in vivo and in vitro to enhance intracellular magnesium and a clinical linkage between cellular magnesium, GSH/GSSG ratios, and tissue glucose metabolism.[8]
Conclusion
Dr. Busby is correct in his recommendation but he does not have intimate experience with the best way to absorb magnesium into the body. The inexpensive pills available in most pharmacies definitely are not a good way of absorbing these minerals. Absorption rates of solid minerals taken in pill form are not nearly as effective as liquid minerals and there is nothing like transdermal magnesium therapy for effective and quick results. Most peoples’ diets are already too high in calcium and too low in magnesium so it is really the magnesium that needs to be stressed, especially in the first months of supplementation. It takes three or four months of heavy transdermal magnesium therapy with magnesium oil to top off cellular levels of magnesium but a year or more if administration is only given orally. Transdermal applications include direct body spraying, medicinal baths as well as magnesium massage.
[2] A radioisotope of an element will bind best to the same substrates which a non-radioactive isotope of the same element will bind. Dr. Stearns has established that when cells are exposed to uranium, the uranium binds to DNA and the cells acquire mutations, triggering a whole slew of protein replication errors, some of which can lead to various cancers. Stearns’ research, published in the journals Mutagenesis and Molecular Carcinogenesis, confirms what many have suspected for some time – that uranium can damage DNA as a heavy metal, independent of its radioactive properties. The biochemical reaction of heavy metals can cause genetic mutations, which in turn can curtail cell growth and cause cancer. Heavy metals that are also radioactive amplify this effect and can cause distortions in shape and thus function even of red blood cells.
[3] http://www.sysbio.org/capabilities/nmr/nih/magnesium.stm
[4] N. A. Littlefield1 , B. S. Hass1, S. J. James1 and L. A. Poirier1(1) U.S. Public Health Service, Food and Drug Administration, National Center for Toxicological Research, Division of Nutritional Toxicology, Department of Health and Human Services, 72079 Jefferson, AR, USA
[5] J Am Coll Cardiol, 2002; 39:695-701. J Am Coll Cardiol, 2002; 39:695-701 American College of Cardiology Foundation.
[6] Virginia Minnich, M. B. Smith, M. J. Brauner, and Philip W. Majerus. Glutathione biosynthesis in human erythrocytes. Department of Internal Medicine, Washington University School of Medicine, J Clin Invest. 1971 March; 50(3): 507–513. Abstract: The two enzymes required for de novo glutathione synthesis, glutamyl cysteine synthetase and glutathione synthetase, have been demonstrated in hemolysates of human erythrocytes. Glutamyl cysteine synthetase requires glutamic acid, cysteine, adenosine triphosphate (ATP), and magnesium ions to form ?-glutamyl cysteine. The activity of this enzyme in hemolysates from 25 normal subjects was 0.43±0.04 ?mole glutamyl cysteine formed per g hemoglobin per min. Glutathione synthetase requires ?-glutamyl cysteine, glycine, ATP, and magnesium ions to form glutathione. The activity of this enzyme in hemolysates from 25 normal subjects was 0.19±0.03 ?mole glutathione formed per g hemoglobin per min. Glutathione synthetase also catalyzes an exchange reaction between glycine and glutathione, but this reaction is not significant under the conditions used for assay of hemolysates. The capacity for erythrocytes to synthesize glutathione exceeds the rate of glutathione turnover by 150-fold, indicating that there is considerable reserve capacity for glutathione synthesis. A patient with erythrocyte glutathione synthetase deficiency has been described. The inability of patients’ extracts to synthesize glutathione is corrected by the addition of pure glutathione synthetase, indicating that there is no inhibitor in the patients’ erythrocytes.
[7] Braverman, E.R. (with Pfeiffer, C.C.)(1987). The healing nutrients within: Facts, findings and new research on amino acids. New Canaan: Keats Publishing
[8] Barbagallo, M. et al. Effects of glutathione on red blood cell intracellular magnesium: relation to glucose metabolism. Hypertension. 1999 Jul;34(1):76-82. Institute of Internal Medicine and Geriatrics, University of Palermo, Italy. mabar@unipa.it
Dr.Sircus is a reader-supported publication. To receive new posts and support my work, consider becoming a free or paid subscriber.
Subscribe now



comments