There are so many reasons why magnesium is essential to health, cancer prevention, and cancer treatment; it would be hard to sum them all up. However, suppose we thread how vital magnesium is to oxygen sufficiency or how magnesium deficiency leads to severe problems in oxygen transport and efficient use of oxygen in the mitochondria. We can then nail down why magnesium is essential in all medical treatments.
Simultaneously, we discover how foolish/stupid/ignorant it is for all branches of medicine to ignore magnesium when it is more critical than all the pharmaceuticals doctors can prescribe. Oncologists who use chemo and radiation but ignore magnesium are committing their patients to much higher incidents of death. Surgeons not using magnesium before, during, and after operations also make surgery more dangerous. Cardiologists who prescribe Statin drugs and ignore magnesium, which most still do, are dooming their patients and societies to extremely high death rates via cardiac arrest.
There is also much evidence that magnesium deficiency, combined with agents that interfere with its utilization, is associated with functional and structural abnormalities of membranes, cells, organs, and systems; you would think that doctors would have figured out that they should highly recommend it.
Besides carbon dioxide (bicarbonates) and oxygen, in terms of importance, magnesium is the next in line of nutrients essential to health and life. It is vital to oxygen but is not thought of that way. It is also crucial to mitochondrial health and function, but most doctors don’t care.
With magnesium deficiency a certainty in all patients, except those who eat spinach three times a day, every day, magnesium is as vital to health recovery as oil is to a car about to go up a mountain range. Getting enough magnesium from our food is almost impossible, and doctors love to prescribe pharmaceuticals that further deplete cellular magnesium levels. And, of course, the stress of modern life puts a nail in the coffin of magnesium sufficiency unless we diligently take our magnesium supplements religiously.
Doctors should know that plasma magnesium levels are unreliable as an index of cellular inadequacy.
Magnesium and the Shape of Red Blood Cells
Red blood cell (RBC) deformability is a crucial factor in determining the movement of red blood cells through microcirculation. The mechanism whereby red cells maintain their biconcave shape has been the subject of numerous studies. One of the critical factors for maintaining biconcave shape is the red cell adenosine triphosphate (ATP) levels. The interaction of calcium, magnesium, and ATP with membrane structural proteins plays a significant role in controlling the shape of human red blood cells.[i]
In healthy people, most red blood cells are smooth-surfaced and concave-shaped with a donut-like appearance. These discocytes have extra membranes in the recessed area that would enable them to move through capillary beds, delivering oxygen, nutrients, and chemical messengers to tissue and removing carbon dioxide and lactic acid.
Abnormal magnesium-deprived red blood cells lack the flexibility to enter tiny capillaries. These nondiscocytes are characterized by irregularities, including surface bumps or ridges, a cup or basin shape, and altered margins instead of the round shape found in discocytes. When people become ill or physically stressed (more magnesium deficient), more discocytes transform into less flexible nondiscocytes. Dr. L.O. Simpson asserts that Fatigue Immune Deficiency Syndrome (CFIDS) results from “Insufficient oxygen availability due to impaired capillary blood flow.”
A biconcave disk is like a donut where the hole doesn’t go all the way through. The biconcave disk shape increases thecell’s surface area, allowing for a greater area for gas exchange.
Intravenous magnesium therapy over 24 hours has increased RBC deformability even in pregnancies with normal RBC deformability. An increase in RBC deformability with magnesium administration offers therapeutic benefits for treating reduced blood flow, as seen in most cases of preeclampsia.[ii]
Magnesium and Hemoglobin
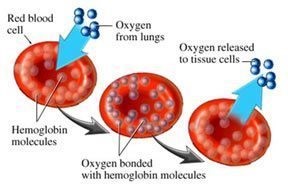
Magnesium enhances the binding of oxygen to haem proteins.[iii] The concentration of Mg2+ in red cells is relatively high, but free Mg2+ is much lower in oxygenated red blood cells than in deoxygenated ones. This suggests some kind of magnesium pump where oxygen climbs aboard the red cells, and magnesium jumps off, only to jump right back on the red cells again.
There is a positive association between serum magnesium and hemoglobin levels in the general population. Of course, the more red blood cells a person has, the more they can carry oxygen. Hemoglobin carries oxygen. The more hemoglobin someone has, the more oxygen they transport. Hemoglobin comprises about a third of the total red blood cell volume. This protein is responsible for transporting more than 98% of the oxygen (the remaining oxygen is dissolved in the blood plasma).
Hypomagnesemia is common and is associated with lower hemoglobin levels in patients with hyperparathyroidism. Hypomagnesemia is common in pre-dialysis CKD patients as well. In this patient group, anemia is independently associated with hypomagnesemia.
Magnesium deficiency is a potent predictor of anemia.
Low iron or iron stores may also cause less oxygen utilization because oxygen binds to iron-containing molecules (heme) in hemoglobin. Low RBC and hemoglobin often reflect an iron deficiency. The prevalence of anemia was 18.3% in men and 31.5% in women. Magnesium and iron intakes were positively associated with hemoglobin levels and inversely related to the prevalence of anemia. The risks of anemia were reduced by 26% and 52%, respectively, for higher iron and magnesium intake. The lowest risk of anemia was observed among participants with the highest intakes of magnesium and iron. Spirulina and chlorella are good sources of both iron and magnesium.
Magnesium and Oxygen Utilization and Healthy Mitochondria
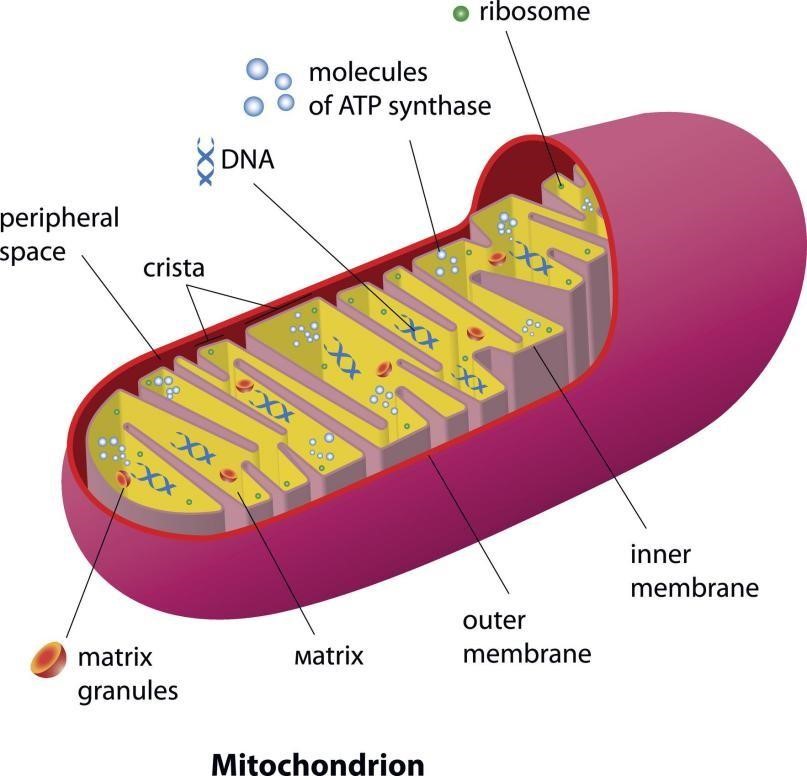
Some medical scientists believe that mitochondrial problems may be at the heart of all chronic diseases, including Cancer. People’s health and energy largely depend on how much oxygen they have and how well their mitochondria utilize it. Dr. Seeger and Dr. Johanna Budwig in Germany have shown that Cancer is mainly the result of faulty energy metabolism in the powerhouses of the cells, the mitochondria. ATP and most of the enzymes involved in energy production require magnesium. A healthy cell has high magnesium and low calcium levels. The problem with low magnesium (Mg) levels is that calcium builds up inside the cells while energy production decreases as the mitochondria gradually calcify.
Magnesium (Mg2+) is critical for all of the energetics of the cells because it is absolutely required that Mg2+ be bound (chelated) by ATP (adenosine triphosphate), the central high-energy compound of the body. ATP without Mg2+ bound cannot create the energy typically used by specific body enzymes to make protein, DNA, RNA, transport sodium or potassium or calcium in and out of cells, nor to phosphorylate proteins in response to hormone signals, etc.
ATP without enough Mg2+ is non-functional and leads to cell death or Cancer. Cells become cancerous exactly when the oxidative process in the mitochondria falters, forcing them to turn to fermentation for survival’s sake. “Bound Mg2+ holds the triphosphate in the correct stereo-chemical position so that it can interact with ATP using enzymes, and the Mg2+ also polarizes the phosphate backbone so that the ‘backside of the phosphorous’ is more positive and susceptible to attack by nucleophilic agents such as hydroxide ion or other negatively charged compounds. Bottom line, Mg2+ at critical concentrations is essential to life,” says Dr. Boyd Haley.
Haley asserts, “All detoxification mechanisms have as the bases of the energy required to remove a toxicant the need for Mg-ATP to drive the process. There is nothing done in the body that does not use energy, and without Mg2+, this energy can neither be made nor used.” Detoxifying carcinogenic chemical poisons is essential for people who want to avoid the ravages of Cancer. The importance of magnesium in cancer prevention and treatment should not be underestimated.
Magnesium and bicarbonate together work to combat the drop in energy within the mitochondria during constant bombardment from toxins. First, magnesium bicarbonate protects the natural organic and inorganic phosphate buffers in the cytoplasm of cells. Second, magnesium bicarbonate neutralizes the acid produced due to metabolic processes and ATP hydrolysis. This allows more ATP to be hydrolyzed or more energy to be made. Magnesium bicarbonate buffers the mitochondria in body cells from excess acid concentrations, improving mitochondrial function and increasing ATP.
If the mitochondria are denied the essential nutrition they need, they cease functioning normally. Dr. Majid Ali says, “Injured mitochondria mutate at much higher rates. Damaged mitochondria are exhausted mitochondria. Exhausted mitochondria cannot produce sufficient ATP molecules. An insufficient supply of ATP molecules means insufficient energy. Insufficient molecular energy means clinical chronic fatigue.”
These organelles are the cell’s power generators, converting oxygen and nutrients into ATP (adenosine triphosphate). ATP is the chemical energy “currency” of the cell that powers the cell’s metabolic activities. This process is called aerobic respiration and is the reason animals breathe oxygen.
Cancer, cardiovascular disease, and neurogenerative diseases, Alzheimer’s being the most common, are all exacerbated dramatically by metabolic disease. They are all metabolic diseases or have their metabolic dysfunction component. And yes, it often starts with magnesium deficiencies.
Magnesium and Inflammation
Scientists think that there’s complex cross-talk between hypoxia and inflammation. According to them, low oxygen may be caused, in part, by cytokines and other inflammatory messengers, which may prevent oxygen from reaching the tissues from capillaries. Many diseases such as heart disease, COPD, asthma, and sleep apnea–involve chronic inflammation. Magnesium as a treatment for inflammation will be covered in a separate chapter.
Magnesium Deficiency and Low Oxygen
“Data shows that magnesium-deficient people use more oxygen during physical activity -their heart rates increased by about ten beats per minute. “When the volunteers were low in magnesium, they needed more energy and oxygen to do low-level activities than when they were in adequate magnesium status.” This is according to a study by physiologist Henry C. Lukaski and nutritionist Forrest H. Nielsen, which reveals essential findings on the effects of depleted body magnesium levels on energy metabolism.
The dissociation of oxygen is also helped by magnesium because it provides an oxygen adsorption isotherm, which is hyperbolic. It also ensures that the oxygen dissociation curves are sigmoidal, which maximizes oxygen saturation with the gaseous pressure of oxygen.[iv]
Oxygen dissociation with increased delivery to the tissues is increased by magnesium by elevation of 2,3-bisphosphoglycerate/DPG (Darley, 1979). Magnesium stabilizes the ability of the porphyrin ring to fluoresce. Free-radical attack of hemoglobin yields ferryl hemoglobin [HbFe4+] (D’Agnillo and Alayash, 2001), which is inhibited by magnesium (Rock et al., 1995).
Conclusion
Magnesium in cancer treatment has a long history (90 years), but oncologists have short memories. Dr. Robert H. Craig, circa 1930, published ‘The Value of Magnesium Chloride as an Aid in the Treatment of Cancer.’ The importance of the biochemical approach to treating cancer with magnesium was also stressed by Prof. Pierre Delbet, Superintendent of the Cancer Institute, Paris. The Stockholm Weekly Journal, back in June 1931, published an article, ‘Take Magnesium and Escape Cancer.’
This chapter focused on magnesium’s crucial role in oxygen transport and utilization and the use of oxygen by the mitochondria because it stabilizes ATP and allows for DNA and RNA transcriptions and repairs.
However, magnesium deficiency is carcinogenic, and in the case of solid tumors, a high level of supplemented magnesium inhibits carcinogenesis.[v] Magnesium repletion has been shown to produce rapid disappearances of the periosteal tumors.[vi]
Magnesium is fundamental to cancer treatment and its avoidance. In Egypt, the cancer rate was only about 10% of that in Europe and America. In the rural fellah, it was practically nonexistent. The main difference was an extremely high magnesium intake of 2.5-3 g in these cancer-free populations, ten times more than in most Western countries.
An inverse relationship between cancer prevalence and the magnesium content of water and soil has been reported in many studies, starting more than 50 years ago. A Russian report showed that stomach cancer is four times more common in the Ukraine, where the magnesium content of soil and drinking water is low, than in Armenia, where the magnesium content is more than twice as high.
A more recent morphologic and statistical analysis of neoplastic deaths in two Polish communities disclosed a nearly three-fold higher death rate in the community with Mg-poor soil than in the one with Mg-rich soil (10%).
[i] http://bloodjournal.hematologylibrary.org/cgi/reprint/44/4/583.pdf
[ii] https://www.informahealthcare.com/doi/abs/10.1081/PRG-45767?cookieSet=1&journalCode=hip
[iii] Terwilliger and Brown, 1993; Takenhiko and Weber; Wood and Dalgleish, 1973
[iv] Murray et al pp. 65-67.
[v] Durlach J, Bara M, Guiet-Bara A, Collery P. Relationship between magnesium, cancer and carcinogenic or anticancer metals. Anticancer Res. 1986 Nov-Dec;6(6):1353-61.
[vi] Hunt, B.J., Belanger, L.F. Localized, multiform, sub-periosteal hyperplasia and generalized osteomyelosclerosis in magnesium-deficient rats. Calcif.Tiss.Res. 1972; 9:17-27.
Dr.Sircus is a reader-supported publication. To receive new posts and support my work, consider becoming a free or paid subscriber.
Subscribe now




comments