Nutritional Status and Viral Disease
The association between viral disease and nutrition has long been thought to be due to effects on the host immune system.[1] This theory suggests that when a host is malnourished, the immune system is compromised, and thus increased susceptibility to viral infection will occur. However, the virus itself may also be affected by the nutritional status of the host. This is true for all viral infections and no less true for Ebola. It is one of the basic reasons that separate the survivors from the majority who perish. The difference between life and death can be measured in the severity of cellular stress.
When it comes to human papilloma virus or HPV, or any virus problem for that matter, it is crucial that you are getting adequate amounts of selenium in your diet or using a supplement. Glutathione requires selenium for its production. Selenium is a trace mineral that the body incorporates into proteins to make over 25 different selenoproteins including the enzyme glutathione peroxidase. Researchers working with the flu virus discovered that animals deficient in selenium were more susceptible to infectious diseases. Animals with a selenium deficiency, when contaminated with the flu virus, the flu virus mutated into a far more virulent form when it was passed on to the next animal. The AIDS infection rate is highest in those African countries with low levels of selenium in their soil.
Selenium holds one of the keys to resolving the killer Cytokine storms that kill Ebola victims.
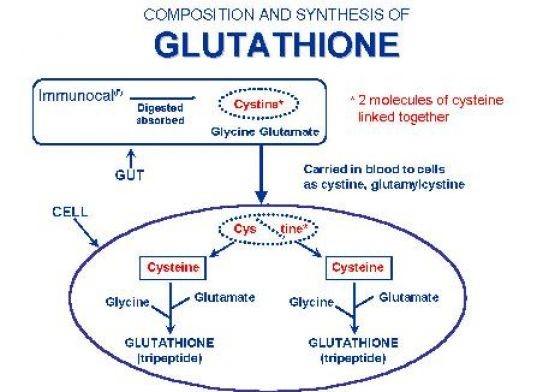
The pre-cursors of glutathione synthesis are the amino acids glutamate, glycine and cysteine, with cysteine being the most important amino acid. As people age or experience disease, glutathione levels in the blood decrease, causing a reduction in this life-sustaining antioxidant. Dietary sources of glutathione are therefore necessary to replenish stores and avoid losses.
Magnesium deficiencies cause glutathione depletion and production of glutathione is also dependent on magnesium.[2] Glutathione synthetase requires-glutamyl cysteine, glycine, ATP, and magnesium ions to form glutathione.[3] According to Dr. Russell Blaylock, low magnesium is associated with dramatic increases in free radical generation as well as glutathione depletion and this is vital since glutathione is one of the few antioxidant molecules known to neutralize mercury. Mercury is included, thanks to deranged organizations like the CDC and the FDA, in peoples’ yearly flu vaccines, and by the ton, is put in population’s mouths in the form of dental amalgam by your local friendly dentist.
Dr. Melinda Beck, a virologist at the University of North Carolina, and Dr. Orville Levander, a nutritional chemist at the USDA’s Agricultural Research Service, described how a run-of-the-mill coxsackievirus mutated into the deadly, rapidly reproducing strain when an infected person or animal was deficient in selenium or vitamin E. When selenium is deficient, we can assume that glutathione levels will be dramatically lower. Dr. E. Will Taylor builds a compelling argument that Ebola contains genes dependent on selenium. Like HIV, when selenium levels in Ebola-infected cells drop, or are low to begin with, the virus reproduces and “escapes” in search of cells with more selenium spreading the infection throughout the body.[4]
The difference is that the genes in the Zaire strain of Ebola genes appear to need 10 times more selenium than does HIV, and Ebola’s greater dependence on selenium may partly account for the speed with which it kills. Normal immune defenses against Ebola would be handicapped if the host-an animal or person-were deficient in selenium. “This raises the possibility that selenium deficiency in host populations may actually foster viral replication, possibly triggering outbreaks and perhaps even facilitating the emergence of more virulent viral strains,” explained Dr. Taylor.
[1] Selenium and host defence towards viruses; Beck MA; Proc Nutr Soc. 1999 Aug;58(3):707-11: http://www.ncbi.nlm.nih.gov/pubmed/10604206
[2] Linus Pauling Institutehttp://lpi.oregonstate.edu/infocenter/minerals/magnesium/index.html#function
[3] Virginia Minnich, M. B. Smith, M. J. Brauner, and Philip W. Majerus. Glutathione biosynthesis in human erythrocytes. Department of Internal Medicine, Washington University School of Medicine, J Clin Invest. 1971 March; 50(3): 507–513
[4] Taylor EW, Antiviral Research, 1995;26:A271-86.
